Graphite Crystal Model STEM Lab
Product Code : SCL-M-12447
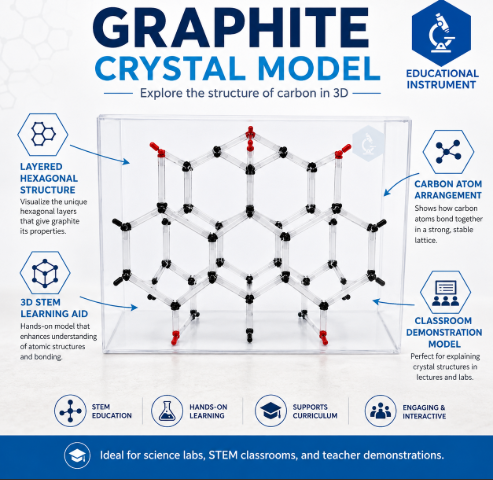
The Graphite Crystal Model by Educational Instrument India is a high-quality, three-dimensional representation of one of the most stable allotropes of carbon. This model is meticulously designed to help students and educators visualize the unique hexagonal layered structure of graphite. Unlike the rigid tetrahedral structure of diamond, this model illustrates the $sp^2$ hybridized carbon atoms arranged in honeycomb sheets, making it an essential tool for understanding solid-state chemistry and materials science.
Crafted with durable, color-coded spheres and high-tension connectors, this model clearly differentiates between the strong covalent bonds within the layers and the weak Van der Waals forces between them. It is an indispensable asset for physics and chemistry laboratories, STEM workshops, and academic institutions.
Technical Specifications
Brand: Educational Instrument India
Material: High-grade Polystyrene (spheres) and resilient Polyvinyl (connectors/rods).
Atomic Configuration: Represents multiple layers (graphene sheets) to demonstrate inter-layer spacing.
Color Coding: Carbon atoms are represented by black spheres; covalent bonds by short grey connectors; Van der Waals forces by elongated rods.
Structure Type: Hexagonal Crystal System.
Dimensions: Scaled for classroom visibility (Standard Lab Size).
Package Contents: 45+ carbon atoms, 50+ covalent bond links, and 12+ inter-layer spacers.
How to Use the Graphite Crystal Model
Assembly: Connect the black carbon spheres using the short grey links to form hexagonal rings (6 atoms per ring).
Layering: Once the honeycomb sheets are formed, use the longer, vertical rods to connect the layers. Note that in graphite, atoms in one layer do not sit directly above atoms in the next (ABAB stacking).
Educational Demonstration: * Conductivity: Use the model to explain how delocalized electrons between layers allow for electrical conductivity.
Lubrication: Demonstrate the sliding motion of the layers to explain why graphite is used as a dry lubricant.
Bonding Analysis: Compare the bond lengths within the hexagon versus the distance between the layers.
Frequently Asked Questions (FAQ)
Q: Why is this model better than 2D diagrams?
A: 3D models provide spatial awareness of the "layer-gap" which is crucial for understanding why graphite is soft and conductive, concepts often lost in 2D drawings.
Q: Is the model pre-assembled?
A: It is usually supplied as a kit to encourage "kinesthetic learning," allowing students to build the lattice from scratch.
Q: Can this model be used for Diamond vs. Graphite comparisons?
A: Yes, Educational Instrument India recommends pairing this with our Diamond Lattice Model to demonstrate carbon polymorphism.


.png)

.png)

.png)

